Working with Mel Science . Chemistry Sets
I worked through the second Mel Science kit received in the mail today, and learned a few lessons about what makes chicken eggs, well, chicken eggs. Read about the first kit here [1].
The kit comes with three experiments to try that can be completed with a single chicken egg. The three experiments are (1) Chemical Omelette (2) Blue Egg White (3) Egg Foam. To begin, a single chicken egg is cracked open and the egg white and yolk are separated. The Mel Science starter kit provides plastic cups and I poured the egg whites into one of them. In retrospect, I should have checked the volume of egg white in a single egg. Each experiment asks for 3 mL of egg white, so a guesstimate of the total volume gives 20 mL or so (?).

Experiment 1 intends to test the chemical reaction egg white protein + Ca(NO3)2 + NaHSO4 –> HNO3 + CaSO4 + NaNO3 + “altered gelly or oil or fat”. This alteration is most visibly observed by a change in color, from the initial white/yellow to a darker yellow with green black splotches.

As I was working under a hood (some might consider this overkill safety precaution for an experiment on eggs, however, safety first always), I did not get a whiff of anything unusual. =)

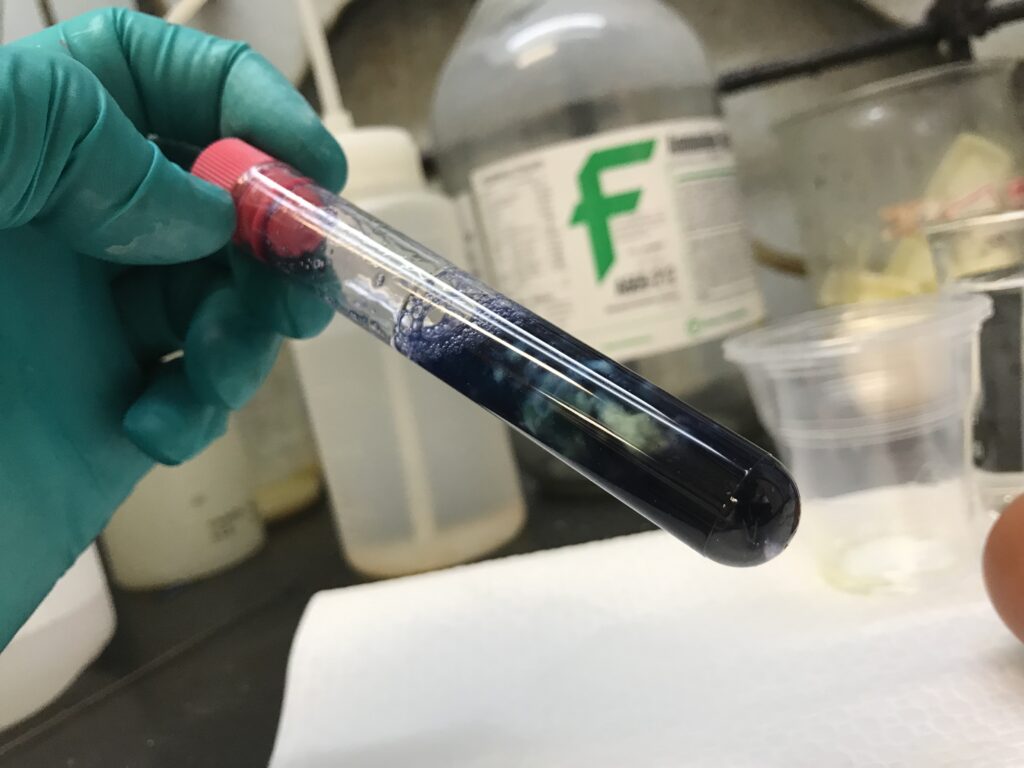
Experiment 2 explores the effects of CuSO4 (solution) and Na2CO3 (whitish powder) on egg white. Water is added to the egg white, and due to differences in density the two materials do not mix (water is lighter [2]) unless aerated by vigorous movement.

CuSO4 (slightly acidic), is added into the egg white + water solution, and immediately stirs up a reaction, see this video here [3] (don’t do this unless medically required). What happens is the even heavier CuSO4 creates strands of “crystals” or “gems” in the surrounding egg white solution. Adding the basic Na2CO3 (approximately 0.2 g) helps bring the pH back to where things started, around 11.
Experiment 3 investigates foaming, creation of gas and the ideal conditions for it’s occurrence (outside the chicken egg). In this particular case the likely gas is relatively odorless CO2, carbon dioxide.
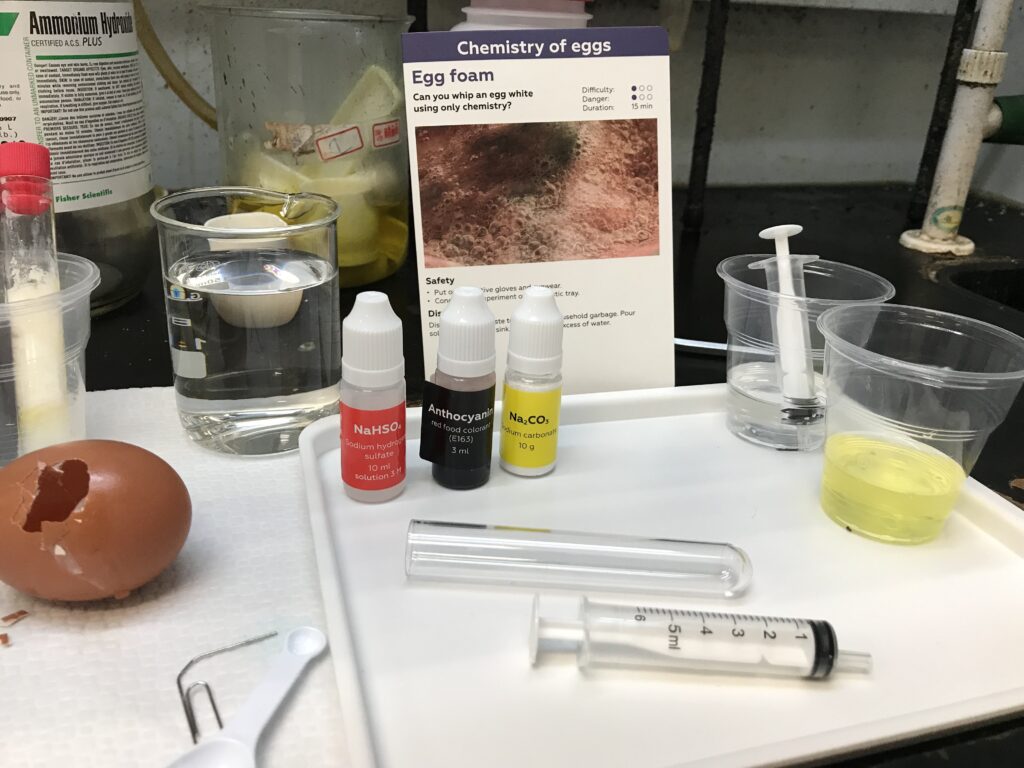
To begin, Na2CO3 is mixed with 3 mL of egg white in a plastic cup, followed by the addition of 10 drops of red food colorant, anthocyanin. Then, 5 mL of NaHSO3 is added into the cup.

A foamy concoction is immediately generated upon stirring [4]. Interestingly, note that the food colorant on bubbles gives a greenish black color (eeks).
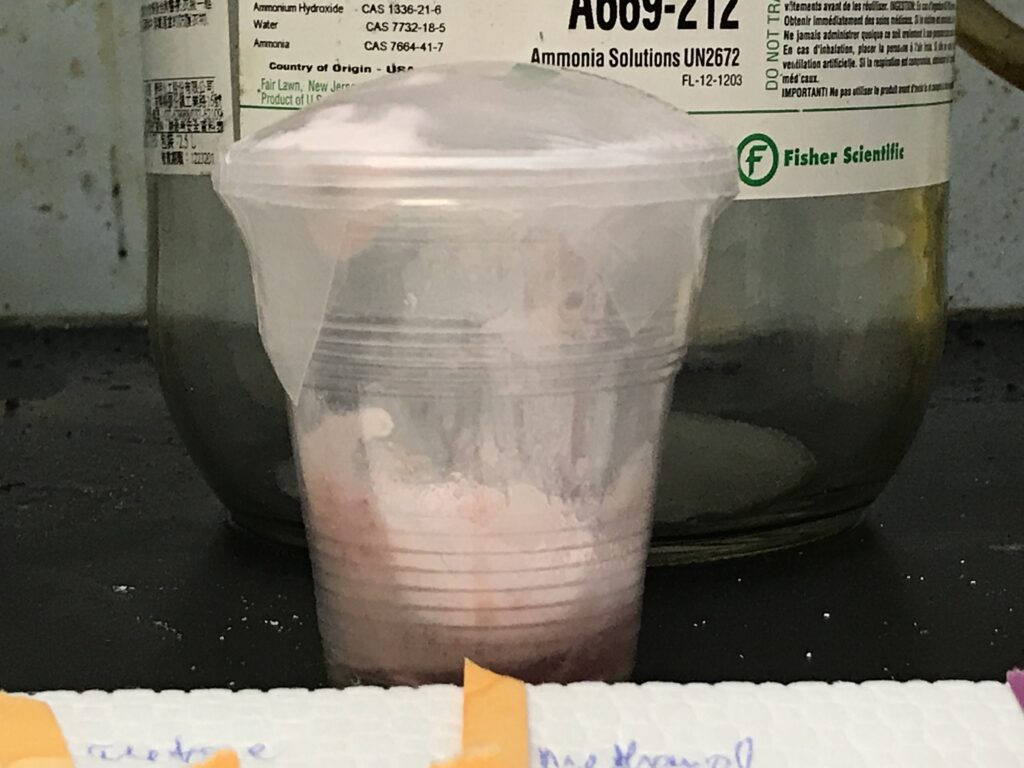
What’s happening is Na2CO3 in NaHSO4 medium leads to dissociation of Na and CO3^2-, and the CO3^2- then breaks down into CO2. The description given with the instructions compares this foamy mixture to meringues, bread and sea foam, although I didn’t try tasting the contents of this cup.
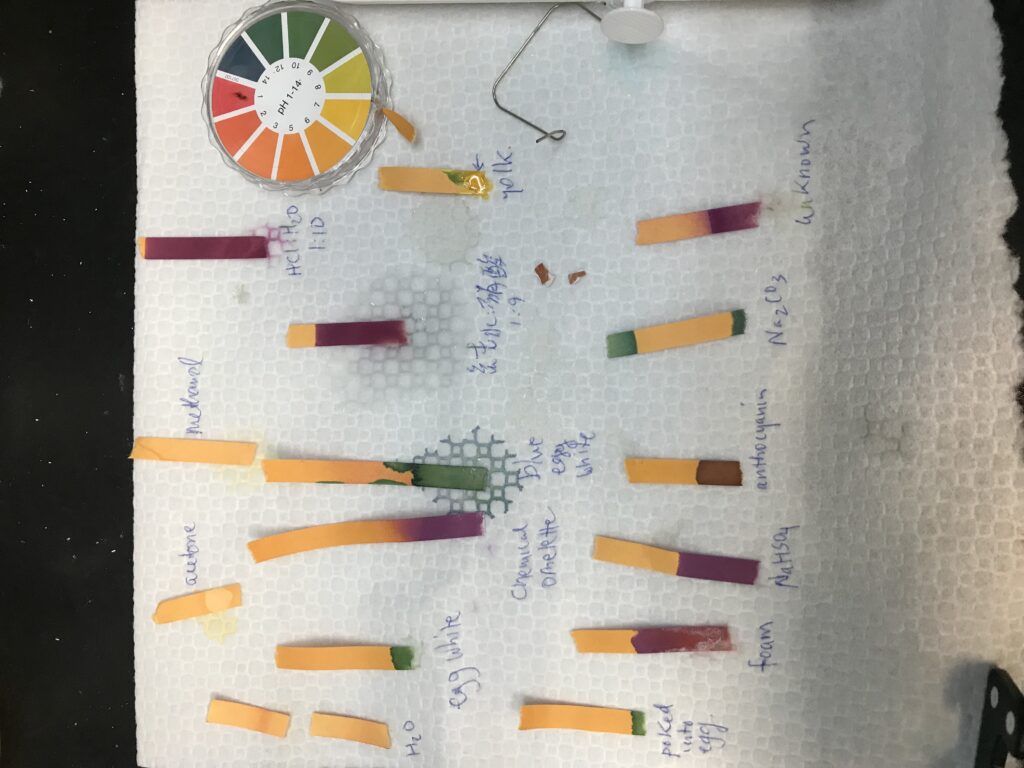
Finally, I found some pH strips, and to wrap up the experiments today, took pH measurements of a few items within reach to create these colorful lines on paper.
References
- https://phillip-wu.com/mel-science-chemistry-tin-錫/
- https://vimeo.com/353380143
- https://www.youtube.com/watch?v=YdpJ0GOU3K4
- https://vimeo.com/353390323